Introduction
This method relates to the adults developmental stage.
The method was developed as a result of discussions within the German Expert Committee on Pesticide Resistance – Insecticides (ECPR-I), and is a modification of a monitoring method formerly used by Bayer Crop Science and Syngenta. It is currently being widely used in Western Europe for monitoring sensitivity of Meligethes spp. populations in oilseed rape to synthetic pyrethroids. Meligethes aeneus is the dominant species of beetle found in European oilseed rape crops.
Materials
- Insect-proof containers
- Fine pointed brush
- Glass beakers for test liquids
- Syringes/pipettes for liquids or weighing balance for solids
- Acetone
- Glass vials (approx. 20ml volume) with lids
- Vial roller (or hotdog roller)
- Small funnel to transfer beetles to vials
- Paper towels
- Ventilated holding cage
- Maximum/minimum thermometer
Method
Collect approximately 100 to 200 adult beetles at different locations across the infested field. Store beetles in an aerated plastic container. Place some dry paper towel at the bottom of the container, and add some oil seed rape leaves plus two or three rape inflorescences as food source (Figure 1). The insects should not be subjected to excessive temperature, humidity or starvation stress after collection. Physical handling of the beetles should be reduced to a minimum.
Use the recording sheet for sampling details and other information that maybe useful for tracking samples and interpreting susceptibility results later on.
Ship the containers as quickly as possible to the test laboratory; transportation method should avoid excessive temperature, humidity or starvation stress
It is recommended that on arrival to the laboratory, the beetles be released into a ventilated holding cage (or equivalent) and left to recover overnight.
The standard test synthetic pyrethroid is lambda-cyhalothrin (technical available from Fluka (or equivalent). Other synthetic pyrethroids can be used, but the vial application concentrations may need to be adjusted to take account of differences in inherent potency between different pyrethroids. It is advisable to initially run a comparison study with lambda-cyhalothrin if an alternative pyrethroid is chosen for your study.
The test containers are glass vials with an internal surface area of 20-80 cm2. Determine the surface area of the glass vials by:
- (h is the height of the vial, r is the radius of the bottom)
- Surface Area = Area of bottom + Area of the side
- Surface Area = π r 2 + (2 π r)* h
Prepare accurate dilutions of the technical grade compound in acetone (p.a). For lambda-cyhalothrin suitable test concentrations in μg per cm2 glass surface have been determined as follows:
- 0.075 µg/cm² (100% of the typical field application rate of 7.5 g ai/ha),
- 0.015 µg/cm² (20% rate),
- Acetone only control
NOTE: Additional rates are required if a full dose response for the generation of accurate susceptibility data (LD50 values) are required.
Glass vials should be filled with 500-1500 μl (depending on vial size, solution should cover base of vial when placed horizontally) of solution and rotated at room temperature until the acetone is completely evaporated (Ensure all acetone vapour is eliminated).
A minimum of two replicates of each concentration and control are required (i.e. 6 vials per test).
NOTE: Additional replicates are required if a full dose response for the generation of accurate susceptibility data (LD50 values) are required.Place between ten and twenty adult beetles per vial (a funnel can be helpful in transferring the beetles to the vial), cap and store at 20 ± 2ºC (Figure 2) and avoiding exposure to an uneven light source or direct sunlight. Ensure that all of vial is equally exposed to light (avoids beetles hiding in refuge of vial cap). Physical contact with the beetles should be kept to a minimum.
The number of beetles severely affected (dead and moribund) is scored after a 24 hour period. The assessment is made by emptying the beetles from the glass vial onto the centre of a piece of paper with a 15cm circle drawn in the middle. The assessment should be made in bright light to stimulate beetle movement out of the circle and the beetles which can not exit the circle before a period of one minute should be considered severely affected.
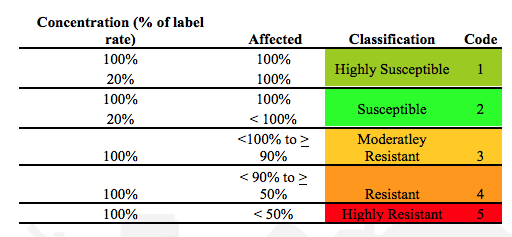
Express results as percentage affected. If greater than 20% of the beetles in the control treatment are severely affected, then the study should be considered as invalid for the purposes of resistance monitoring
Using the following ‘susceptibility rating scheme’ evaluate the test population as being in one of the following categories: highly susceptible, susceptible, moderately resistant, resistant and highly resistant.
Precautions & Notes
- Where glass equipment is used it must be adequately cleaned with an appropriate organic solvent before re-use to prevent cross-contamination.
- Different batches of technical grade insecticide may vary in concentration of active ingredient (usually between 85-99% a.i.). It is recommended to use high purity a.i. where possible. Purity needs to be taken into account when preparing the test solutions.
Comments
Sample recording and assessment sheets are available by downloading the pdf file of Method No: 011